SHARE
Services for the Health in Asian & African Regions (SHARE) = SHARE is a citizen sector organization (NGO) that engages in international cooperation mainly through providing health service.
HOME > News > Information of COVID-19(6)
- 【※updated】COVID-19(multi-language)
- Information of COVID-19(15)
- Information of COVID-19(vaccines)
- 《Request to the Government of Japan》Ease the protection of intellectual property rights and promote sharing and cooperating on pharmaceuticals and medical technologies, in order to accelerate efforts to overcome COVID-19 all over the world.
- Information of COVID-19(14)
- Information of COVID-19(13)
- Information of COVID-19(12)
- Information of COVID-19(11)
- SHARE Medical Information Line for Migrants is resumed in Oct.
- Information of COVID-19(10)
Information of COVID-19(6)

Information of COVID-19 (6)by SHARE 2020.05.11
1.Global and Japanese Situation of COVID-19
Confirmed cases of COVID-19 worldwide according to reports by WHO Update 2020/5/7 (Japan Standard Time) :Ministry of Health, Labor and Wellfare (MHLW)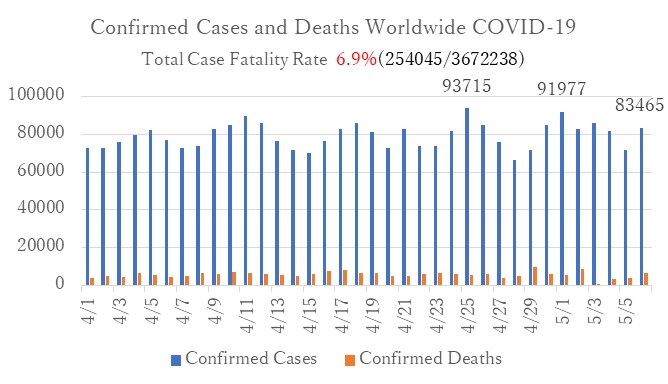
- Globally, the number of daily confirmed cases continues inreasing by more than 80,000.
- In Europe, while the number of daily confirmed cases decreased in Italy, France, and Germany within last month; Russia's daily confirmed cases is exceeding 10,000.
Tokyo 2020/5/07 (Japan Standard Time): Tokyo Prefecture Report

- The number of daily confirmed cases is decreasing nationwide as well as in the Tokyo metropolitan area. This might be an impact of staying home under the state of emergency. However, careful surveillance is necessary during these two weeks following Golden Week. Therefore, the Japanese government has extended the state of emergency until the end of the month.
2.Change definition of close contacts
- On April 20, MLHW changed definition of transmission period of COVID-19 from "the day of onset" to "2 days before the appearance of symptoms".
- The definition of close contacts includes these people as follows:
- Have lived together, or had long hours contact with the patients (e.g., in the train or aircraft).
- Have examined, nursed or cared for the patients without appropriate personal protective equipment.
- Have a high possibility of directly contacting airway secretions or body fluids of patients.
- Have been in contact with the patient for more than 15 minutes without any necessary infection prevention measures (mask, etc.) within a touchable distance (1 meter as a guide).
Reference:
https://www.niid.go.jp/niid/ja/diseases/ka/corona-virus/2019-ncov/2484-idsc/9357-2019-ncov-02.html
3.New coronavirus testing guidelines
- Up until now, the confirmed cases have been treated at hospitals. Hospitals must keep available beds for severe cases as the number of confirmed cases is increasing. Therefore, the mild case patient will be transferred to the designated facilities.
- On May 8, the MLHW eased access to the testing of COVID-19 infection.
- The new guidelines said that those who need to seek assistance immediately, and to call to a local health center, are as follows:
- Those who have one of symptoms such as difficulty in breathing, strong fatigue or high fever.
- Senior citizens, persons with underlying health conditions, and pregnant women who had mild cold symptoms.
- Those who have mild cold symptoms more than four days.
Reference:
https://www.mhlw.go.jp/content/000628620.pdf
4.Potential treatments and vaccines for the COVID-19
- Vaccination is one of the most effective ways to prevent disease. It helps the body's immune system to recognize pathogens like viruses, which keeps us safe from diseases. Therapeutic medicine is to treat after the symptoms appear, prevent worsen, and avoid death from the communicable disease.
- Human testing of investigational drugs consists of three phases.
- Phase1 to establish a drug's safety involving healthy volunteers,
- Phase 2 to assess the drug's effectiveness involving patient volunteers,
- Phase 3 to determine the effectiveness and identify other side effects by applying more number of patients
This process needs to be carried out ethically. - WHO says 70 vaccines in the works, with three leading candidates as of April 13
- The furthest along in the clinical process is developed by Hong Kong-listed CanSino Biologics Inc. and the Beijing Institute of Biotechnology, which is in phase 2.
- The second is mRNA-1273 (RNA vaccine) developed by Moderna Inc and US National Institute of Health (NIH), which is in phase1.
- The third is INO-4800 (DNA vaccine) developed by Inovio Pharmaceutical Inc., which is in phase1. - Progress is occurring at with unprecedented speed in the development process, and the drug industry is hoping to compress the time to get a vaccine to market to within the next year.
- Currently, there is no therapeutic medicine which has been confirmed in its effectiveness scientifically to treat COVID-19. 109 clinical trials in the works, with leading candidates "Remdesivir" and "Favipiravir."
- Remdesivir was previously tested as an Ebola treatment. It has generated promising results in animal studies for MERS and SARS, which are also caused by coronaviruses, suggesting its effect in patients with COVID-19.
- Clinical trial in China reported on April 29 that there was no association of clinical benefits (e.g., faster time to clinical improvements) by utilizing Remdesivir (Wang, 2020).
- US NIH reported on the same date that Remdesivir accelerates time to recover and suggested decreasing mortality rate.
- On May 7, MLHW granted the Special Approval for Emergency on Remedisivir for the treatment of patients with COVID-19.
- Favipiraivir (Avigan), is a drug approved as an anti-influenza drug in Japan in 2014. It selectively and potentially inhibits the RNA polymerase (RdRp) of RNA viruses. It demonstrated anti-viral activity against Ebola virus (Furuta, 2017).
- Favipiravir is under clinical trials in Japan. However, the side effects on fetal anomaly limit its use on pregnant women and women expecting to become pregnant.
- Currently, there is no therapeutic medicine that has been shown to be safe and effective. Over 100 countries are working together to find effective therapeutics under solidarity clinical trial for COVID-19 launched by the WHO.
(Vaccine)
(Therapeutic medicine)
Reference: ・https://www.drugs.com/fda-approval-process.html
・https://www.bloomberg.com/news/articles/2020-04-13/who-says-70-vaccines-in-the-works-with-three-leading-candidates
Wang Y. et al., Remdesivir in adults with severe COVID-19: a randomized, double-blind, placebo-controlled, multicenter trial. Lancet, Published Online April 29, 2020.
・https://www.niaid.nih.gov/news-events/nih-clinical-trial-shows-remdesivir-accelerates-recovery-advanced-covid-19
・https://www.pmda.go.jp/english/int-activities/0004.pdf
Furuta Y. et. al., Favipiravir (T-705), a broad spectrum inhibitor of viral RNA polymerase. Proc Jpn Acad ser B Phys Biol Sci. 2017; 93(7): 449-463.
・https://www.who.int/emergencies/diseases/novel-coronavirus-2019/global-research-on-novel-coronavirus-2019-ncov/solidarity-clinical-trial-for-covid-19-treatments
Written by Nakasa T, MD, Co-President of SHARE
Koto K, RN, SHARE advisor
Koto K, RN, SHARE advisor
■English Information
-
Information of COVID-19(1)(ENGLISH)
-
Information of COVID-19(2)(ENGLISH)
-
Information of COVID-19(3)(ENGLISH)
-
Information of COVID-19(4)(ENGLISH)
-
Information of COVID-19(5)(ENGLISH)
-
Information of COVID-19(6)(ENGLISH)
-
Information of COVID-19(7)(ENGLISH)
-
Information of COVID-19(8)(ENGLISH)
-
Information of COVID-19(9)(ENGLISH)
■Useful URLs by multi-languages




